-
Exclusives
-
About
9-12 High School
This easy and inexpensive lab demonstrates the transfer of heat energy from a higher temperature substance to a lower temperature substance within the same system, establishing the second law of thermodynamics and the concept of thermal equilibrium. Schedule it as a class demonstration or as a small group activity.
Students generate temperature data, graph collected temperature data, and use the data to establish and explain the concepts of heat transfer and thermal equilibrium. After completing the data analysis, students use energy diagrams to visualize and explain changes in heat energy for each component of the system.
Ask students to generate a list of instances when 2 substances come together and the temperatures of both substances change. Identify the direction the thermal energy is traveling. Begin with the examples of butter melting on pancakes and ice melting in hot tea. Compile a class list and leave it available for students to see. They will use the list during the assessment portion of the activity.

How can evidence of heat transfer between components of a system be measured?
PE HS-PS3-4. Plan and conduct an investigation to provide evidence that the transfer of thermal energy when two components of different temperatures are combines within a closed system results in a more uniform energy distribution among the components in the system (second law of thermodynamics).
Analyzing and Interpreting Data
PS2.A: Forces and Motion
Cause and Effect
Students need to have proper eye protection for working with warm water and hand protection for pouring the warm water into a graduated cylinder and cup. Remind students of the proper use of thermometers.
The day before the activity, prepare a 50 mL ice cube for a class demonstration or a cube for each student group. Prior to the activity, warm enough water to supply the demonstration or each group with 50 ml of water. It should be approximately 30 to 40° C above room temperature.
No disposal is required. Calorimeters may be saved for additional activities. Preparing the cardboard tops for the calorimeter cups prior to the lab will save time.
A. Construction of a closed system (calorimeter)
B. System 1
C. System 2
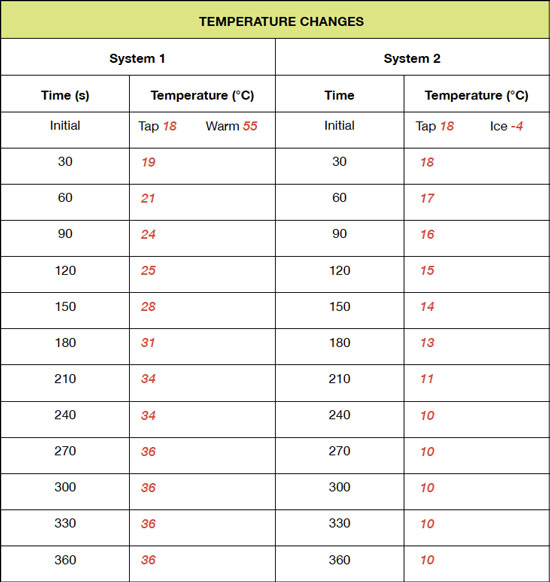
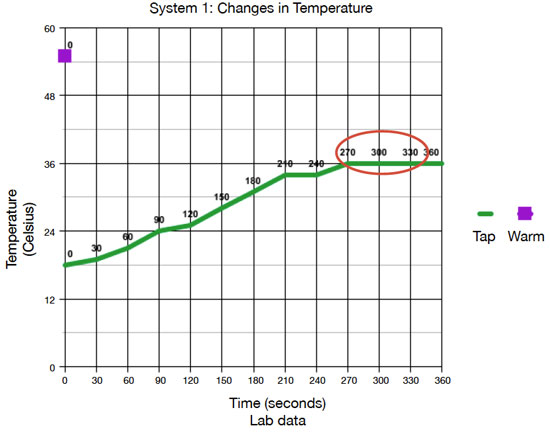
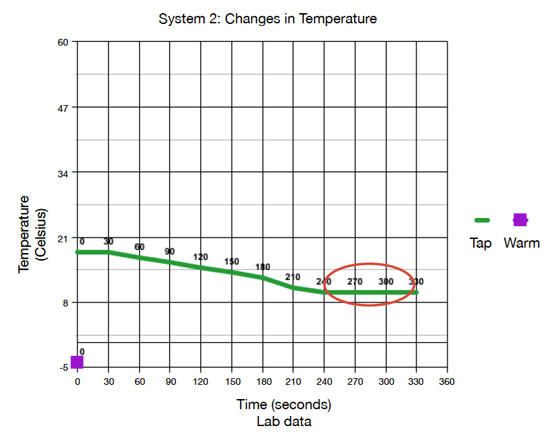
Define the system, its initial state, and final state for each experiment.
System 1 is the 50 mL of tap water initially at 18° C and the 50 mL of warm water initially at 55° C, which is contained by the calorimeter or foam cups. The thermometer interacted with the water in the system. The final state of the system at thermal equilibrium is 100 mL of water at 36° C. System 2 is the 50 mL of tap water initially at 18° C and the 50 mL ice cube initially at -4° C, which is contained by the calorimeter or foam cups. The thermometer interacted with the water in the system. The final state of the system at thermal equilibrium is 100 mL of water at 10° C.
For each system, explain in which direction heat flowed. Use your data to support your claim.
In system 1, heat flowed from the warmer water to the tap water. The data show that the temperature of the tap water increased, so thermal energy from the warmer water flowed into the tap water to warm it. In system 2, thermal water flowed from the tap water into the ice cube to warm/melt it. This is supported by the temperature of the tap water decreasing while the ice cube melted, indicating that the ice cube temperature increased.
On each graph, circle the location in which thermal equilibrium is reached.
See the graphs above.
On a molecular level, explain how thermal equilibrium is obtained.
The substance at the higher temperature has more kinetic energy (KE) than the substance at the lower temperature. As particles collide, kinetic energy is transferred from the substance with more kinetic energy to the substance with less kinetic energy. This process continues until all particles have about the same kinetic energy, which is defined as thermal equilibrium.
Use a series of energy diagrams to visualize the 2 processes of energy transfer in this activity.

*Next Generation Science Standards® is a registered trademark of Achieve. Neither Achieve nor the lead states and partners that developed the Next Generation Science Standards were involved in the production of this product, and do not endorse it.
Carolina is teamed with teachers and continually provides valuable resources–articles, activities, and how-to videos–to help teachers in their classroom.
Get the latest news, free activities, teacher tips, product info, and more delivered to your inbox.