From Jellyfish to the Bench
Green fluorescent protein (GFP) is used in teaching biotechnology, genetics, and AP® Biology. What is the significance of this protein other than allowing students to create fluorescent green bacteria? Perhaps your students have seen news headlines about transgenic animals that fluoresce green under ultraviolet (UV) light because the gene for GFP has been inserted into their genome. Students may also have seen fish at the pet store that glow green for the same reason. However, few students are probably familiar with the less sensationalistic uses of GFP in science.
The discovery and structure of GFP
Aequorea victoria is a jellyfish found off the Northwest Coast of the United States and is known for its luminescence. In 1962 Osamu Shimomura and his colleagues isolated the luminescent substance from animals they collected at Friday Harbor in Washington. The substance turned out to be 2 proteins, aequorin and the now well-known GFP.Since that time, much has been learned about GFP. In A. victoria, GFP is located with aequorin in granules around the edge of the jellyfish’s bell. When aequorin binds calcium ions in the lab the protein releases energy as blue light. However, rather than emitting blue light in the animal, the aequorin protein transfers the energy to GFP. This transferred energy then causes GFP to fluoresce a yellowish green. In the lab, GFP does not require aequorin to fluoresce; light of the appropriate wavelength (blue or near UV) will also cause the protein to fluoresce. A barrel-shaped structure within the protein is responsible for the fluorescence; the barrel is composed of 11 beta sheets with an alpha helix in its center.
What about GFP makes it a powerful research tool?
GFP and mutant versions of it are very useful for scientists studying the inner workings of cells and organisms. Scientists introduce GFP into cells by inserting its gene into the genome of the cell or organism being studied. The following characteristics make GFP a useful tool for studying cells and organisms:Relatively nontoxic to cellsStable over a broad pH rangeResistant to heat and detergentsContinues to fluoresce even after being stimulated for a relatively long periodDoes not require any cofactors to fluoresceTo make it even more amenable for use in research, scientists created many mutated versions of the protein. For example, the original GFP protein does not fold well at 37° C; however, many of the cells and organisms that scientists study function at 37° C. Therefore, one of the first mutations made altered the protein so that it would fold well at 37° C. Mutations have also been created that increase its fluorescence. Furthermore, GFP has been mutated to create variants that fluoresce in a surprising array of colors. For the remainder of this article, “GFP” will be used to refer to the original protein and its variants.
The first use of GFP was as a reporter protein
The first use of GFP as a research tool was when its gene was used as a reporter gene in the nematode worm, Caenorhabditis elegans. A reporter gene is a gene that is used to detect when a promoter is activated. The reporter gene is placed after the promoter of interest. When the promoter is turned on, the reporter protein is made, and in the case of GFP, green fluorescence is detected. Because GFP is not highly toxic to cells and doesn’t require any toxic substrates, it can be used to detect when a specific promoter is turned on in living cells or a living organism.
Using GFP to localize proteins in a cell
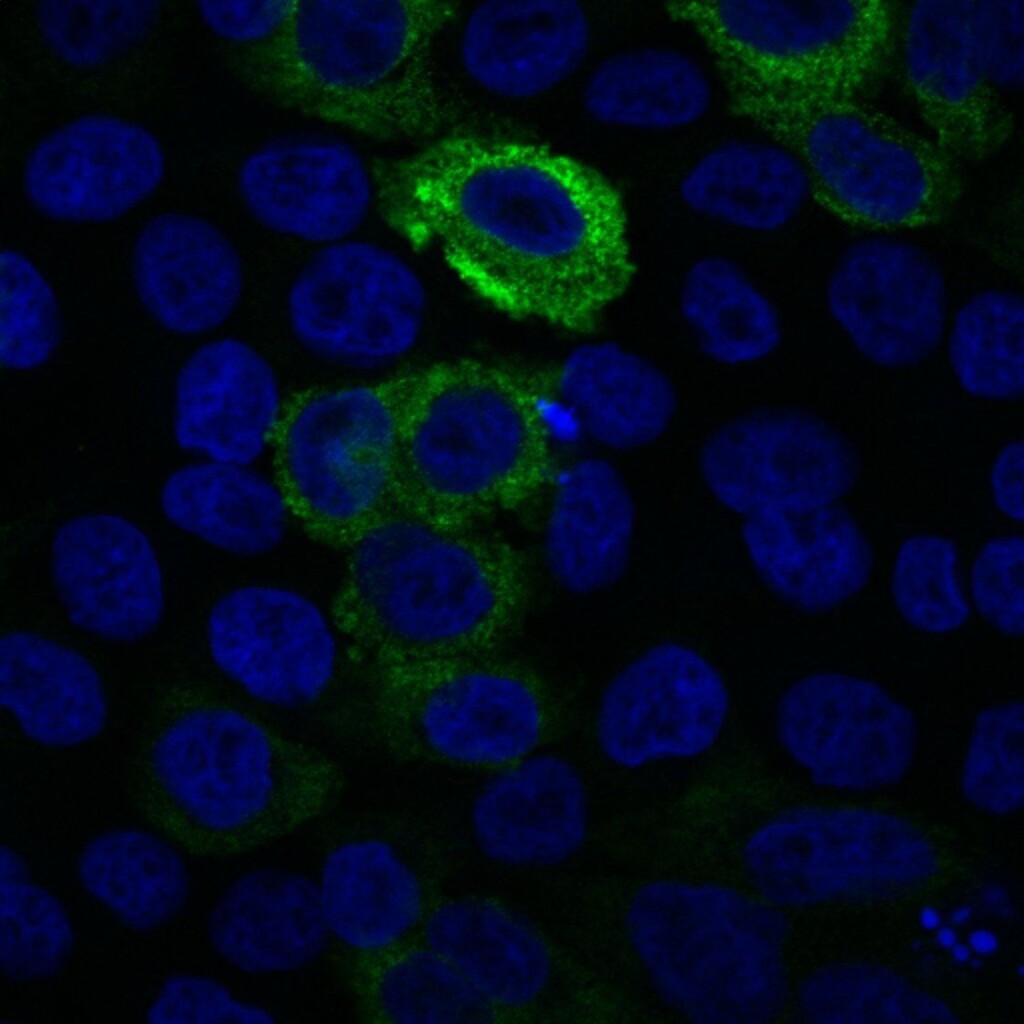
Protein, a protein that contains both GFP and the protein of interest. To do this, the gene for GFP is linked to the gene for the protein of interest, and the fused gene inserted into the genome of the cell or organism being studied. GFP has generally been found to not interfere with the folding of the protein or partial protein amino acid sequence it is fused to. In addition, GFP usually does not appear to interfere with the normal function or movement of the protein it is fused to.The location of the protein the GFP is fused to can be determined by illuminating the cells under a microscope with light at a wavelength that will cause the GFP fusion protein in the cells to fluoresce. Wherever the protein is in the cell, there will be spots of bright fluorescent green (or other color depending upon the GFP used). This same technique can be used to determine where 2 proteins are located in relationship to each other. Fusion proteins can be created for each of 2 proteins, but a different color of GFP is used for each protein. Thus, the 2 proteins can be visualized in the same cell.GFP provides an advantage over older techniques used for localizing specific proteins. In the past, studies localizing proteins were performed using antibodies specific for the protein(s) of interest linked to traditional fluorescent molecules (those not coded for by genes). The procedures used for visualizing proteins with these antibodies kill the cells. In contrast, the use of GFP allows cells to be kept alive over the course of the experiment. Thus, movement of the proteins can be tracked over time.
GFP can be used to determine the location of a protein in a cell by creating a fusion
Using GFP to study signal transduction
GFP and its variants are increasingly being used to study the signaling pathways cells use to sense their environment and to respond to what they sense. The proteins and other molecules that constitute these pathways interact in very specific ways in response to various external and internal stimuli. Understanding these interactions helps scientists better understand how a cell functions, and also facilitates drug development. Some of the interactions between proteins in these pathways occur rapidly and are difficult to track. A technique called fluorescence resonance energy transfer (FRET) is used to detect close interaction between 2 molecules. In FRET, the proteins whose interaction is being studied are each labeled with a different fluorescent molecule (i.e., different colors of GFP). When the proteins are in close proximity, the fluorescent molecules interact with each other. The interaction alters the fluorescence of each in a way that can be measured.
GFP as a sensor
GFP molecules can also be used as sensors. For example, many studies in cell biology involve detecting the pH or pH change in an organelle or in a certain location of a cell. Mutated GFP molecules have been created that change the wavelength that they maximally absorb at or that cease to fluoresce in response to specific pH changes. A fusion protein can be created that links such a GFP molecule to a specific protein sequence (a tag) that targets the protein to a specific location in the cell.In the past, indicator molecules that were used to detect pH and pH changes were often toxic to the cell, and the methods used to get the molecules into the cell were invasive and disruptive to normal cell function. When GFP is used, once the gene expressing GFP or the GFP fusion protein is introduced into the cell, no other cofactor or substrate must be introduced. Thus, no invasive procedures must be performed to introduce any additional molecules into the cell. This makes the experiment easier to perform and the data more reliable.
Using GFP to study development
The study of the development of organisms has been facilitated by the use of GFP. Much of development involves the migration of cells to their final location in the organism. In the past, scientists studied cell migration by examining nonliving embryos or cross sections of embryos at various stages of development, and deducing the major events by the order in which they occur. Developments in microscopy have made it possible, in some situations, to track development in live samples. To do this, cells, organelles, or proteins are “tagged” so that a cell’s location within the organism can be tracked.GFP can be used to do the tagging by introducing the gene coding for GFP or a GFP fusion protein into the organism’s genome. Which cell type the GFP or GFP fusion protein is expressed in can be determined by the choice of the promoter used to express the gene. Changes in the location of cells within an organism can then be tracked over time by observing the location of the fluorescence.
Using GFP to study cancer metastasis
Scientists also use GFP to study cancer metastasis. To do so, they first introduce the gene for GFP into cancer cells that are cultured in the lab. The GFP-expressing cancer cells are then introduced into an experimental animal, such as a mouse. Tumors, primary as well as metastatic, form in the mouse. These tumors and cancer cells will fluoresce when exposed to the appropriate wavelength of light, and thus can easily be tracked and studied. In some situations and using the right equipment, tumors can even be examined in the intact, live animal.Additional uses of GFP not covered in this article also exist and have opened up new areas of investigation in many fields of research. Moreover, other similarly useful fluorescent proteins have been isolated from other organisms, including coral, sea anemones, sea squirts, and sea pens. If your students are using GFP in a lab, consider having them research these other fluorescent proteins and the additional uses of GFP. It is a good way to increase their understanding and appreciation of this protein that has so dramatically impacted scientists’ ability to study many of the major questions in biology.
References
Campbell, T.N. and Choy, F.Y.M. (2001) The Effect of pH on Green Fluorescent Protein: a Brief Review. Molecular Biology Today. Vol. 2(1), 1—4.
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W.W., Prasher, D.C. (1994) Green Fluorescent Protein as a Marker for Gene Expression. Science. Vol. 263, 802—805.
Hoffman, R.M. (2015) Application of GFP Imaging in Cancer. Laboratory Investigation. Vol. 95, 432—452.
Mehta, S. and Zhang. J. (2011) Reporting from the Field: Genetically Encoded Fluorescent Reporters Uncover Signaling Dynamic in Living Biological Systems. Annual Review of Biochemistry. Vol. 80, 375—401.
Nowotschin, S. and Hadjantonakis, A. (2014) Live Imaging Mouse Embryonic Development: Seeing Is Believing and Revealing. Methods in Molecular Biology. Vol. 1092, 405—420.
Shimomura, O. (2005) The Discovery of Aequorin and Green Fluorescent Protein. Journal of Microscopy. Vol. 217, Pt I, 3—15.
Tsien, R.Y. (1998) The Green Fluorescent Protein. Annual Review of Biochemistry. Vol. 67, 509—544.
Zimmer, M. (2002) Green Fluorescent Protein (GFP): Applications, Structure, and Related Photophysical Behavior. Chemical Reviews. Vol. 102, 759—781.
About The Author
Carolina Staff
Carolina is teamed with teachers and continually provides valuable resources–articles, activities, and how-to videos–to help teachers in their classroom.