Common mistakes you may be making in your biotechnology lab
Have you ever dedicated days to prep and procedure only to find out you have mixed results or–even worse–no results at all?
Biotechnology is an exacting science. One small mistake can have major effects on downstream results. We are here to help with you with the dos and don’ts that will set you up for success in your biotechnology classroom.
Bacterial Transformation Dos and Don’ts
Bacterial transformation lab is a multiday procedure that takes much prep work.
1. Proper media preparations. DO NOT add antibiotics to media when it is too hot. Doing this can degrade the antibiotics, which can yield variable results. Always follow the directions in your teacher’s manual on how to prepare your media.
You should allow the agar to cool until the bottle can be held in a bare hand (although the agar will still feel very warm–around 55 °C).
2. Pick the right one(s). DO pick an isolated bacterial colony 2—3 mm in diameter or isolated colonies that equal 2—5 mm in diameter. This ensures the cells are at the optimal growth phase yielding a high transformation efficiency.
3. That’s shocking! The heat shock is an integral step in bacterial transformation. DO make certain that your water bath is set at 42 °C and follow the procedure outlined in your teacher’s manual. After the incubation period, DO make sure your transformation tube immediately goes into your ice bucket.

Electrophoresis Dos and Don’ts
Electrophoresis is a foundational biotechnology technique. Running, staining, and destaining the perfect gel takes practice. But with that practice, and adhering to our protocols, you can run beautiful gels every time!

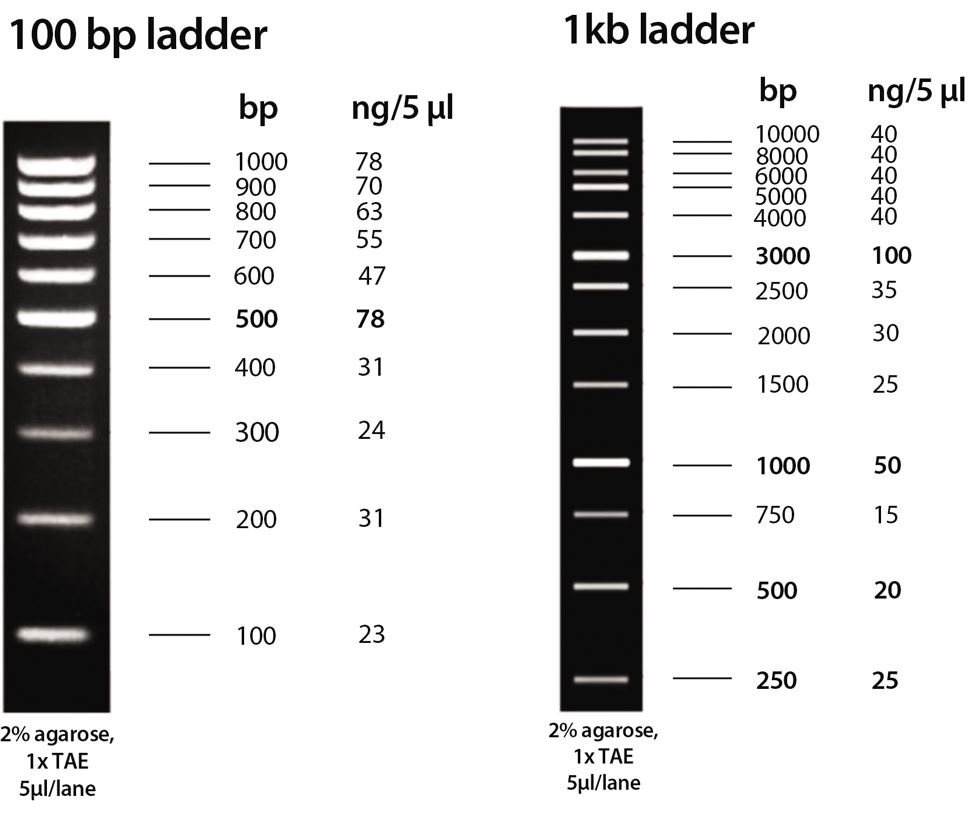
1. Controls, controls, controls. If your kit comes with a molecular (weight) marker (DNA ladder), DO use this as a reference point. The DNA ladder not only acts as a size marker but also can tell you a lot about your electrophoresis experiment. The absence or presence of your marker points to a failed or successful polymerase chain reaction (PCR), staining process, or gel makeup.
2. “Franken-gel.” DO NOT use reagents and supplies from different sources with our kits. Using parts and pieces from different kits requires time to troubleshoot and optimize and doesn’t necessarily mean you will yield good, reproducible results. Our kits are written and optimized to work with all the reagents provided, saving you time and hassle.
3. What if my gel doesn’t look anything like the picture in my teacher’s manual? There are many variables that affect the banding pattern of a gel. Please see our Field Guide to Electrophoresis Effects for more dos and don’ts.

4. Follow directions for stains. DO read allthe staining and destaining protocols in your teacher’s manuals. We use several different nucleic acid stains, which are optimized to the included reagents and DNA samples. This means the protocols are not necessarily interchangeable.
Polymerase Chain Reaction Dos and Don’ts
Carolina was the first science education company to offer polymerase chain reaction (PCR) kits to classroom teachers. We are experts when it comes to helping you get better, more reproducible results.
Our PCR kits involve several techniques. One kit can involve a combination of molecular techniques. These techniques and their results build on one another and, therefore, rely on close attention to protocol. If one mistake is made during a technique upstream of the protocol, it can affect the entire experiment. For these reasons, the following pitfalls incorporate dos and don’ts for both electrophoresis and for PCR experiments.
As long as you’re confident in the viability of your DNA sample, you can reasonably conclude that a failed PCR experiment is due to the PCR itself, or the gel electrophoresis.
1. Again, controls. If your PCR kit comes with a molecular (weight) marker (DNA ladder), DO use this as a reference point. The DNA ladder not only acts as a size marker but can tell you a lot about your PCR experiment. The absence or presence of your marker points to a failed or successful PCR, staining process, or gel makeup.
2. Use the kit. DO NOT use reagents and supplies from different sources with our kits. Using parts and pieces from different kits requires time to troubleshoot and optimize and doesn’t necessarily mean you will get good, reproducible results.
3. It’s all in the programming. DO program your thermal cycler using the PCR steps in the teacher’s manual. Our developers have fine-tuned and tested this program. We know it works, and if you follow the provided steps, it will work in your classroom too.

Did you have a failed PCR experiment and don’t know where it went wrong? Try these helpful tips to troubleshoot.
There can be many reasons for failed PCR experiments. The following explanations are designed to help you reason through your results and determine possible scenarios. They are not inclusive of all possibilities.
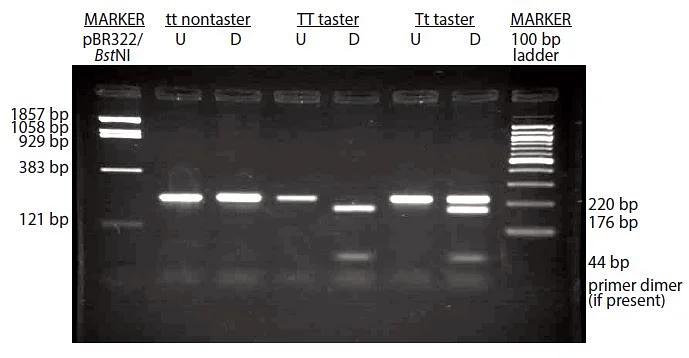
Result: There are no bands at all.
If your gel has no bands whatsoever (see below), including the molecular marker, this suggests a problem with the makeup of your gel. Make certain the gel is made with the appropriate buffer and agarose.
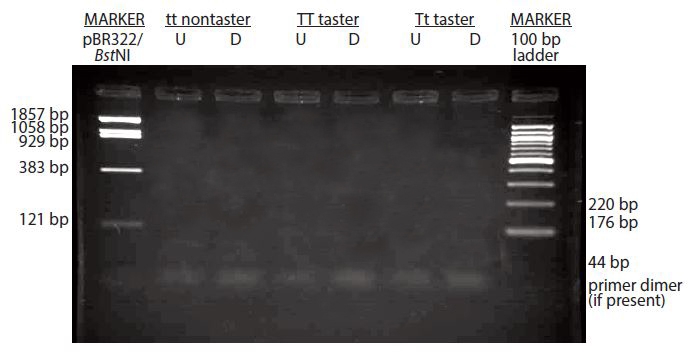
Depending on your method of staining, if there is a lot of background noise, perhaps you need to fully destain your gel to visualize the bands.
Result: The molecular marker is present, but there are no other bands.
If your gel shows the molecular marker but no bands from your samples, this points to a problem with the isolation or amplification of the DNA. The reason for this explanation is the molecular marker is a known DNA sequence that has been digested with restriction enzymes. It is often quality controlled and is not used in the lab flow of the experiment of your unknown DNA samples. It can be loaded onto a gel and visualized without the isolation/amplification/digestion steps. The marker not only determines the size of your bands but also acts as a control.
We’re Here to Help
- Need help troubleshooting your PCR kit? Send an image of your gel to product@carolina.com.
- For more tips and other useful info about your biotechnology kits, contact us.
- Get a list of our PCR offerings and browse PCR equipment.
Additional Reading: Biotechnology: An Essential Curriculum




