
-
Exclusives
-
About
In this lab activity students use a simple dichotomous key to identify several genera of algae contained in our algae mixtures. The key avoids the use of highly technical terms so that even beginning students can use it.
Determine stream water quality based on a macroinvertebrate inventory.
This LabSheet can be used with your choice of the following Algae Mixtures. The key does not cover the 151221 Volvocales Mixture or the 151216 Algae Survey Mixture as these have their own keys.

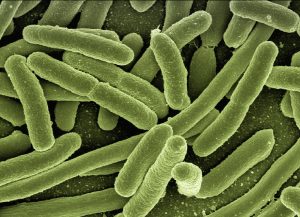
This Bacillus subtilis culture is Gram+, spore-forming, motile rods, and an antibiotic producer.
Ensure that students understand and adhere to safe laboratory practices when performing any activity in the classroom or lab. Demonstrate the protocol for correctly using the instruments and materials necessary to complete the activities, and emphasize the importance of proper usage. Use personal protective equipment such as safety glasses or goggles, gloves, and aprons) when appropriate. Model proper laboratory safety practices for your students and require them to adhere to all laboratory safety rules. None of the algae included in these mixtures is parasitic or pathogenic. Even so, know and follow your district’s guidelines so you are prepared if a student should ingest a culture. Cultures remaining after the completion of the activities can be flushed down a sink with tap water. The chlorine and chloramines in most tap water will kill the algae. If your tap water is not chlorinated, pipet 1 mL of bleach (sodium hypochlorite) or isopropanol (rubbing alcohol) into the culture and wait 15 minutes before flushing down the sink.
When you receive your cultures, remove them from the shipping container. Loosen and remove the lids. Use the dropping pipets included with your order to aerate the culture. Use a separate pipet for each culture. After aeration, replace the lids loosely on the jars but do not screw them down. Place the cultures in a window or under a light bank until ready to use. Use the cultures within 7 days of receipt if possible. Algae mixtures are not suitable for long-term culturing. For continuous culturing of algae, use our individual algae cultures.
Students can work individually or in groups of 2.
Set up workstations for each mixture. Each station should have the following:
Algae Mixture
dropping pipet
microscope slides
coverslips
concavity slides (needed for Volvox only)
Optional:
Students can be assigned to give currently acceptable classifications for the algae they observed. They can also make tables in which they record their observations of the different algae.
*Next Generation Science Standards® is a registered trademark of Achieve. Neither Achieve nor the lead states and partners that developed the Next Generation Science Standards were involved in the production of this product, and do not endorse it.















Get the latest news, free activities, teacher tips, product info, and more delivered to your inbox.