-
Exclusives
-
About
9-12 High School
This activity explores the phenomenon of chemical precipitation and asks students to construct an atomic level model of precipitation using ionic and net ionic equations. Initially, students run a series of reactions. Some reactions produce precipitates and some don’t. Using only the reactions that produce precipitates, students then write ionic equations, cross out spectator ions, and conclude with the net ionic equation. The activity may be used as an extension to the reference kit, Mystery Chemical Reactions or as a stand-alone visual introduction to precipitates, solubility, ionic and net ionic equations.
How is the phenomenon of precipitate formation explained on the atomic level?
HS-PS1-7. Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction.
Analyzing and Interpreting Data
PS1.B Chemical Reactions
Patterns
Use safety goggles, gloves, and apron. Wash hands with soap and water when finished. Use this kit only in accordance with established laboratory safety practices, including appropriate personal protective equipment (PPE) such as gloves, chemical splash goggles, and lab coats or aprons. Ensure that students understand and adhere to these practices. Students should not eat, drink, or chew gum in the lab and should wash their hands before or after entering and exiting the lab. Because they might irritate or burn the skin, avoid contact with the dilute solutions in this lab. If contact occurs, flush the affected area with water.
Solutions in dropper bottles may be stored for additional classes or additional activities. Know and follow all federal, state, and local regulations as well as school district guidelines for the disposal of laboratory wastes.
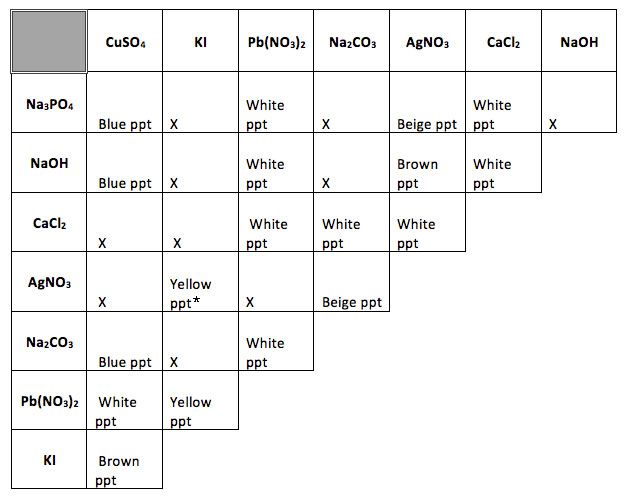
*If the silver nitrate solution is old and the silver appears tarnished, silver precipitate colors may vary slightly.
Identify all reactions that produced a precipitate and write a balanced chemical reaction to model the bonds being broken and reformed at the atomic level and conservation of matter.
See below. Note the (ppt) designation may be written as (s)
What evidence do you have that all the reactants are soluble?
All the reactant solutions were transparent and showed no signs of the reactant falling out of solution as a solid.
Using the chart of Solubility Rules above, identify the product that is the precipitate and place a (ppt) or (s) to the right of the product formula. Place an (aq) to the right of all chemicals that are soluble.

Convert the balanced chemical equation to an ionic equation to model the process of dissociation. Split apart all soluble chemicals into a cation and anion. Show the charge on the ion. If needed, change the coefficient to reflect the total number of ions in solution.
For example:
2 Ca(NO3)2 (aq)→ 2Ca2+ (aq) + 4NO3-(aq)

With a single line, mark out the spectator ions with the coefficients. Write the net ionic equation modeling the formation of the precipitate on the atomic level. Make certain it is balanced to illustrate conservation of matter. See above for the strikethroughs.

Use an ionic and net ionic equation to explain why equations are not written for reactions in this activity that do not produce precipitates
For reactions in this activity that do not produce a precipitate, all ions remain in the aqueous state. No molecular compounds are formed either. Consequently, all ions get canceled and there is no net ionic equation that can be written.
An example equation follows:
NaOH(aq) + KI(aq) NaI(aq) + KOH(aq)
Na1+(aq) + OH1-(aq) + K1+(aq) + I1-(aq) Na1+(aq) + I1-(aq) + K1+(aq) + OH1-(aq)
No net ionic equation possible. All ions are spectator ions and are crossed out.
*Next Generation Science Standards® is a registered trademark of Achieve. Neither Achieve nor the lead states and partners that developed the Next Generation Science Standards were involved in the production of this product, and do not endorse it.
Carolina is teamed with teachers and continually provides valuable resources–articles, activities, and how-to videos–to help teachers in their classroom.
Get the latest news, free activities, teacher tips, product info, and more delivered to your inbox.